A wide variety of targets are included, but most contain oxygen functional groups, and relatively few contain other heteroatoms such as nitrogen and sulfur. Of this page you can practise devising syntheses of relatively simple target molecules using the retrosynthetic analysis strategy. Many useful resources are freely available on the web the following links are good starting points. The process is much more complex that than this brief description suggests for more details consult textbooks or your course notes. Then analyse the subtarget in the same way, and continue in this fashion until you arrive at readily available starting materials. In retrosynthetic analysis, you start with the target and work backwards, asking yourself "What could this target be prepared from, using a reliable reaction?" to arrive at a 'subtarget'. Corey at Harvard University and is called 'retrosynthetic analysis'. The principal method for devising synthetic routes was developed by Prof E.J.
To meet that demand, chemists have devised principles that allow them to design synthetic routes for the preparation of just about any 'target' compound. There is a never-ending demand for new, better, chemical products to sustain and improve our lives, e.g medicines, polymers, dyes, food additives, etc. The synthesis of useful substances is, arguably, the most important activity of chemists. This data is being shared through a google account – and the associated Google Drive.On completing these problems you should be able to:ĭevise syntheses of simple targets using the principles of retrosynthetic analysis.ĭemonstrate a knowledge of synthetically important carbon-carbon bond forming reactions and functional group interconversions.

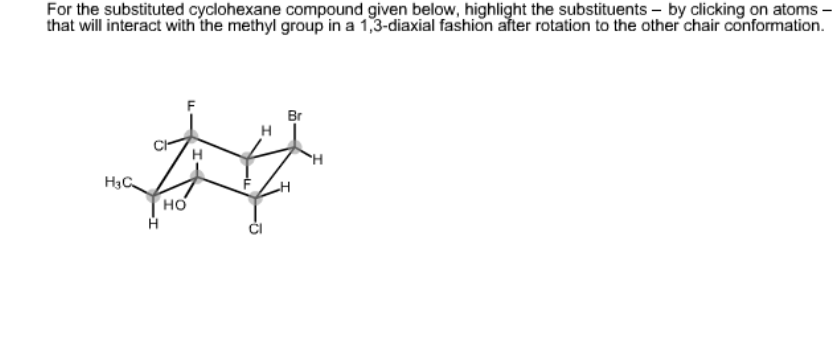
CYCLOHEXANE IN CHEMDOODLE SOFTWARE
Gaussian and GaussView are well-established computational chemistry software packages.molecular orbitals and orbital energies.vibrational frequencies and vibrational modes.heats of formation and other thermodynamic properties.MolCalc is a web interface that allows chemist to build small molecules and estimate molecular properties such as.So, below are some recommended resources for some computational chemistry that are useful when considering computational thermochemistry. Computational chemistry like ab initio electronic structure computation has become an invaluable resource for physical chemists wanting to make the connection between theory and experiment. You can do 10’s or 100’s of computer ‘experiments’ in much less time than you can physical experiments. I also highly recommend doing computational chemistry to calculate heats of reaction (i.e., combustion, formation) BEFORE doing any experiments. In my opinion, it is very very useful to simulate bomb calorimetry before actually doing it in person.įrom simulations and even just looking about basic information about bomb calorimetry instrumentation, a student can PREDICT how much of a temperature change is expected BEFORE actually doing the experiment.
I would love to see this simulation be updated for modern HTML5 similar to PHET.
